| Item No.: | 21936-01 |
|---|---|
| Manufacturer item no.: | 500-STR-DE-10 |
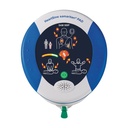
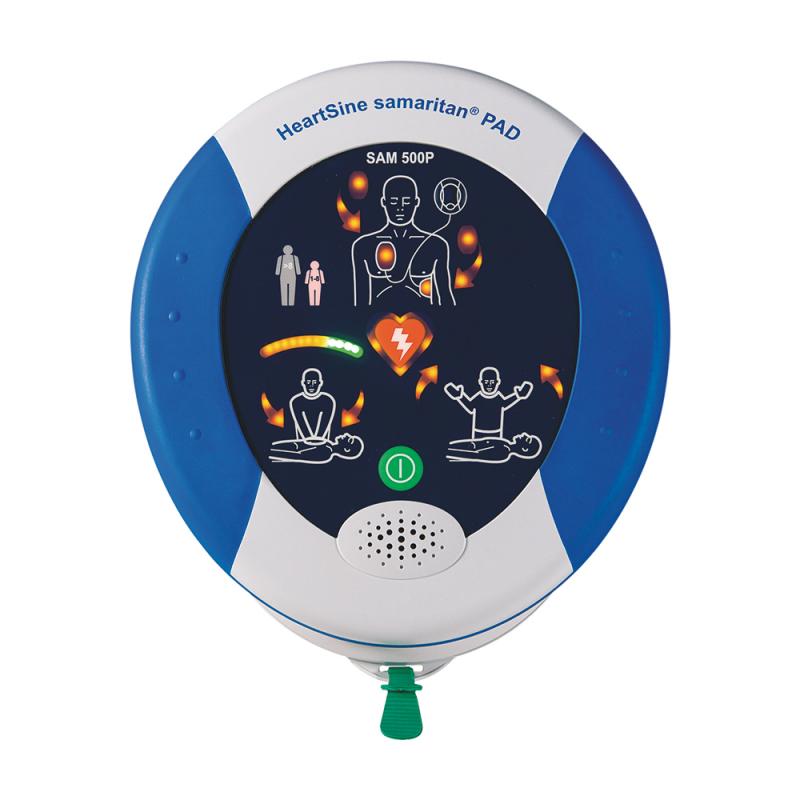
Easiest operation thanks to 1-button function
Compact design, lightweight at 1.1 kg
Extremely robust with IP56 rating
Escalating energy protocol 150 J, 150 J, 200 J
Low-maintenance with 4-year interval and exempt from STK
CPR Advisory/Feedback to support resuscitation quality, visual and audible
Scope of delivery:
- 4-year battery and electrode cassette (Pad-Pak™) for adults and children > 8 years or > 25 kg (only one expiry date!)
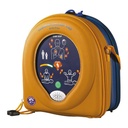
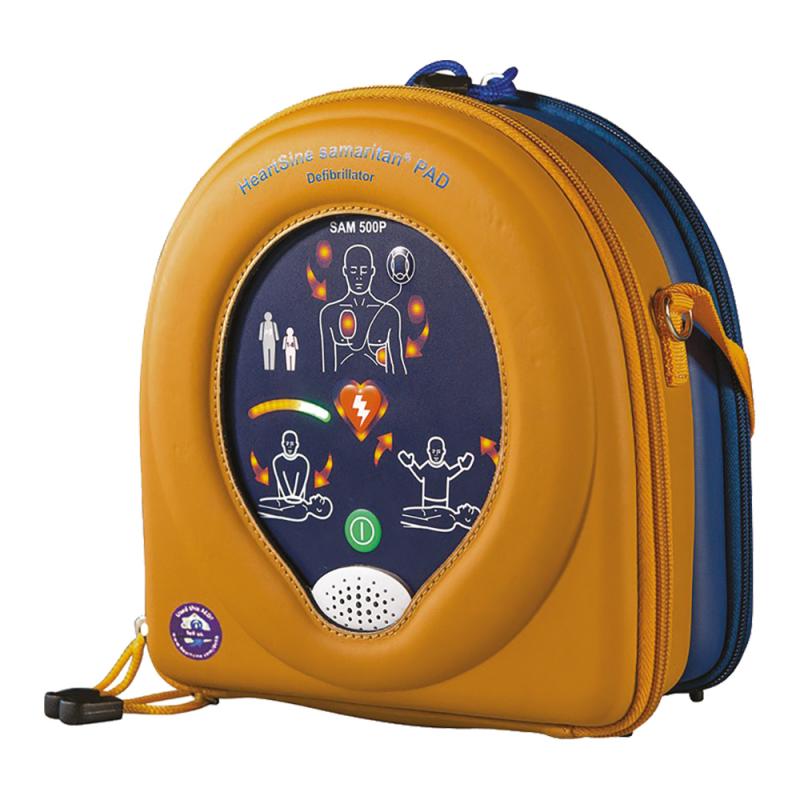
- Multi-function double carry bag, yellow/blue
- Instructions for use, quick guide
- Certificates and approvals CE 0120, FDA, UL, ISO, STK exempt, IP56, aviation approval
- Updatable with new AHA / ERC / Illcor guidelines
Kontaktinformationen im Rahmen der Produktinformationsverordnung (GPSR)
Gesetzliche Anbietererkennung:
HeartSine Technologies, Ltd.
207 Airport Road West, BT3 9ED, Belfast, Nordirland
Mailkontakt:
Product portfolio
HeartSine primarily develops and manufactures AED devices from the “samaritan® PAD” series for public access defibrillation (“Public Access Defibrillators”). These devices are compact, easy to use, robust and designed to enable effective use by both laypersons and first responders in cases of sudden cardiac arrest. Many models offer integrated visual and audible instructions as well as features such as CPR rate feedback and Wi-Fi connectivity for device monitoring and readiness checks.