| Item No.: | 22459-01 |
|---|---|
| Manufacturer item no.: | 672032 |
| PZN: | 15616304 |
| Item No.: | 22459-02 |
|---|---|
| Manufacturer item no.: | 672042 |
| PZN: | 182076 |
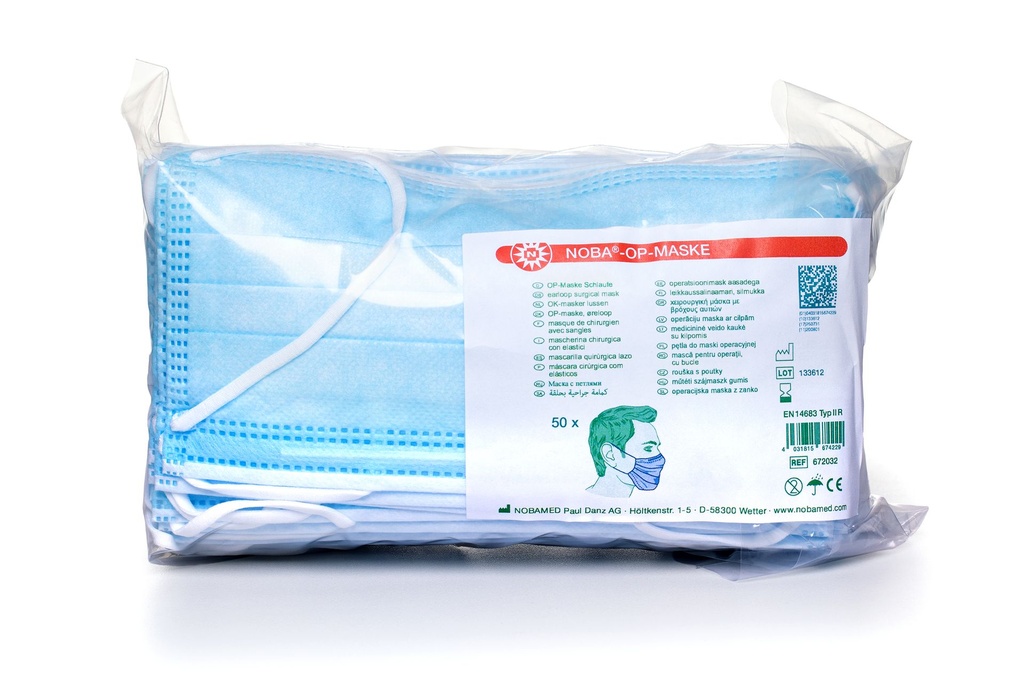
✓ Filtration efficiency: > 98%
✓ meets the requirements of DIN EN 14683, Type II R
✓ Reliable protection for patients
✓ For users: protection against splashes of contaminated liquids
✓ Fibreglass- and latex-free
3-ply, hypoallergenic medical face mask with elastic
ear loops, ≥ 98% filtration efficiency and a very low differential pressure. All
masks have very high bacterial filtration efficiency, thereby providing
reliable protection for the patient.
For the user, additional
protection against splashes of contaminated liquids is provided. The masks have
integrated nose clips that can be adjusted to the bridge of the nose and
are characterised by extra-soft finished edges and inner fleece. The masks
are fibreglass- and latex-free.
Composition:
Polypropylene, latex-free elastomer thread,
Nose clip made of stainless steel
Normative and legal requirements:
NOBA® surgical mask is a medical device
in accordance with the German Medical Devices Act (MPG), Directive 93/42/EEC and
Regulation MDR (EU) 2017/745 and
is classified as Class I, Rule 1.
It meets the requirements of DIN EN
14683, Type II R ("Medical face masks, requirements and test methods").
The product contains no hazardous
toxic substances in accordance with REACH.
It bears CE and DIN EN ISO 15223-1
labelling at all packaging levels.
Kontaktinformationen im Rahmen der Produktinformationsverordnung (GPSR)
Gesetzliche Anbietererkennung:
NOBAMED Paul Danz AG
Höltkenstraße 1-5, 58300, Wetter (Ruhr), Deutschland
Mailkontakt: shop@nobamed-ag.com