| Item No.: | 23212-01 |
|---|---|
| Manufacturer item no.: | 09417125702 |
• It is intended for testing individuals with suspected COVID-19 or with known or suspected exposure to SARS-CoV-2.
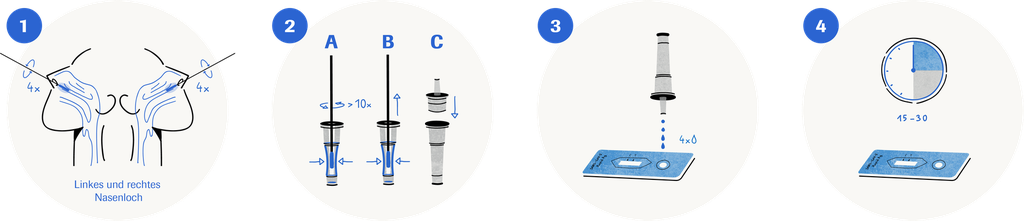
• For nasal sample collection, the specimen is taken from the front part of the nose and not from the nasopharyngeal cavity. This sampling method is less invasive and therefore more comfortable for the patient.
• In clinical studies, the SARS-CoV-2 Rapid Antigen Test Nasal showed, for professionally collected samples, a relative sensitivity of 90.6% (Ct value ≤ 30; 95% CI: 75.0%–98.0%) and a specificity of 98.6%. For self-collected samples under professional supervision, a sensitivity of 84.4% (Ct value ≤ 30; 95% CI: 67.2%–94.7%) and a specificity of 99.2% were determined. Overall, the studies included 468 symptomatic and asymptomatic individuals.
Product data:
Assay format: Lateral flow test / immunochromatographic (in vitro test)
Test type: Qualitative
Instrument: No instrument required
Sample material: Nasal
Target antigen: Nucleocapsid (N)
Read-out time: 15–30 minutes
Storage temperature: 2–30 °C
Sensitivity Ct ≤ 30
Collection by healthcare professional: 90.6%
Self-collection: 84.4%
Specificity
Collection by healthcare professional: 98.6%
Self-collection: 99.2%
Impact of mutations: Based on current knowledge, there is no impact on performance due to variants, e.g. from England and South Africa.
Impact of mRNA vaccine: No impact; the Pfizer and Moderna vaccines both use the spike protein and not the nucleocapsid (N) protein.
Kontaktinformationen im Rahmen der Produktinformationsverordnung (GPSR)
Gesetzliche Anbietererkennung:
Roche Deutschland Holding GmbH
Emil-Barell-Str. 1, 79639, Grenzach-Wyhlen, Deutschland
Mailkontakt: grenzach.communications@roche.com

Products from Roche
Roche offers innovative diagnostic solutions and therapies worldwide for the prevention, detection and treatment of diseases – with a strong focus on research, personalised healthcare solutions and sustainable medical care.