| Item No.: | 18493-01 |
|---|---|
| Manufacturer item no.: | CU-SP1 |
| EAN: | 8023279353402 |
Adult electrodes, disposable battery, carry bag, user manual
Product description:
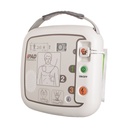
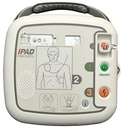
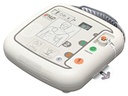
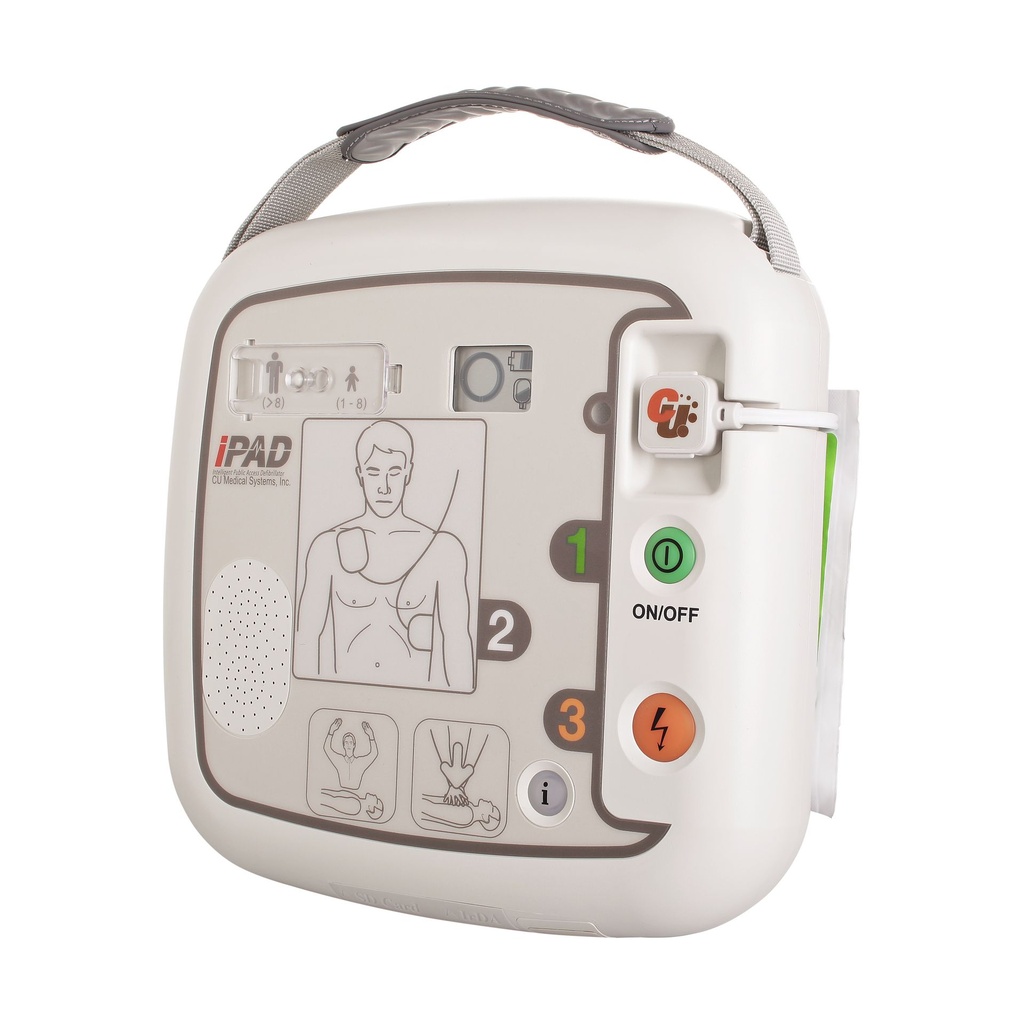
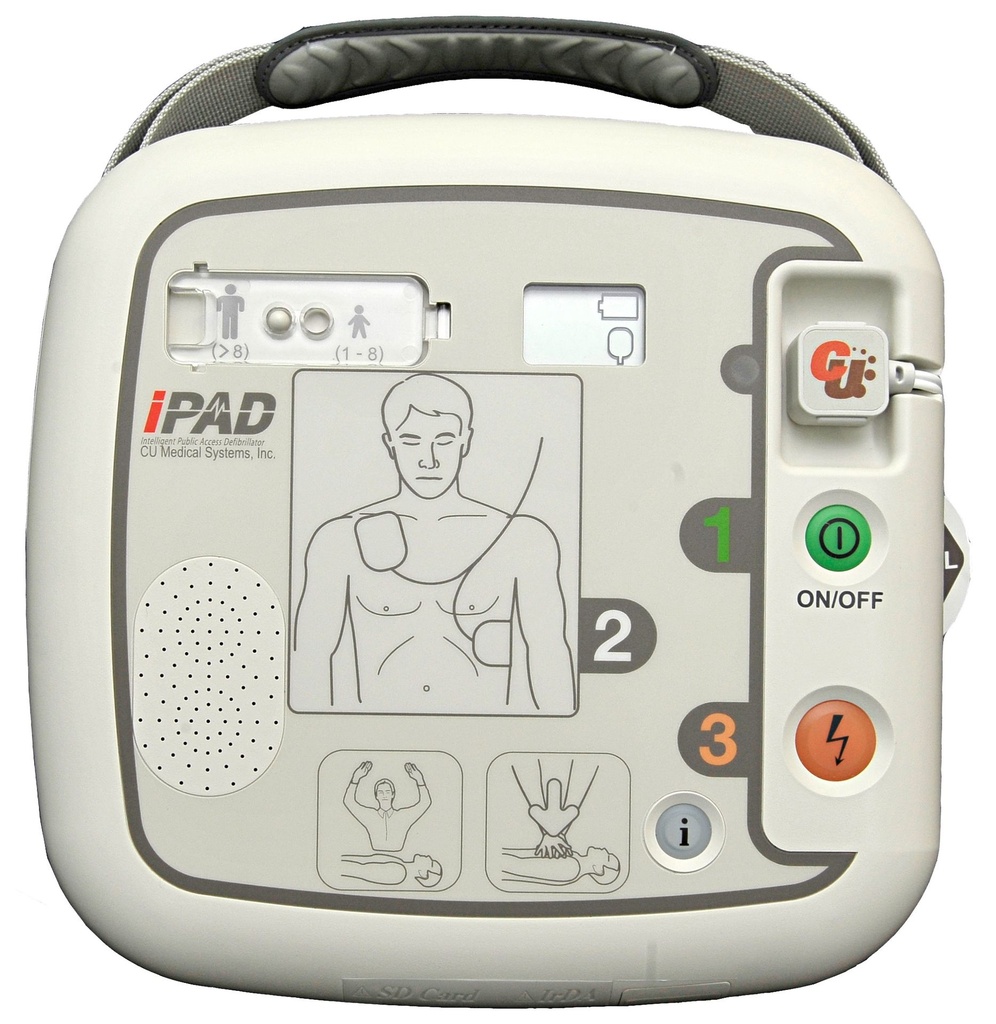
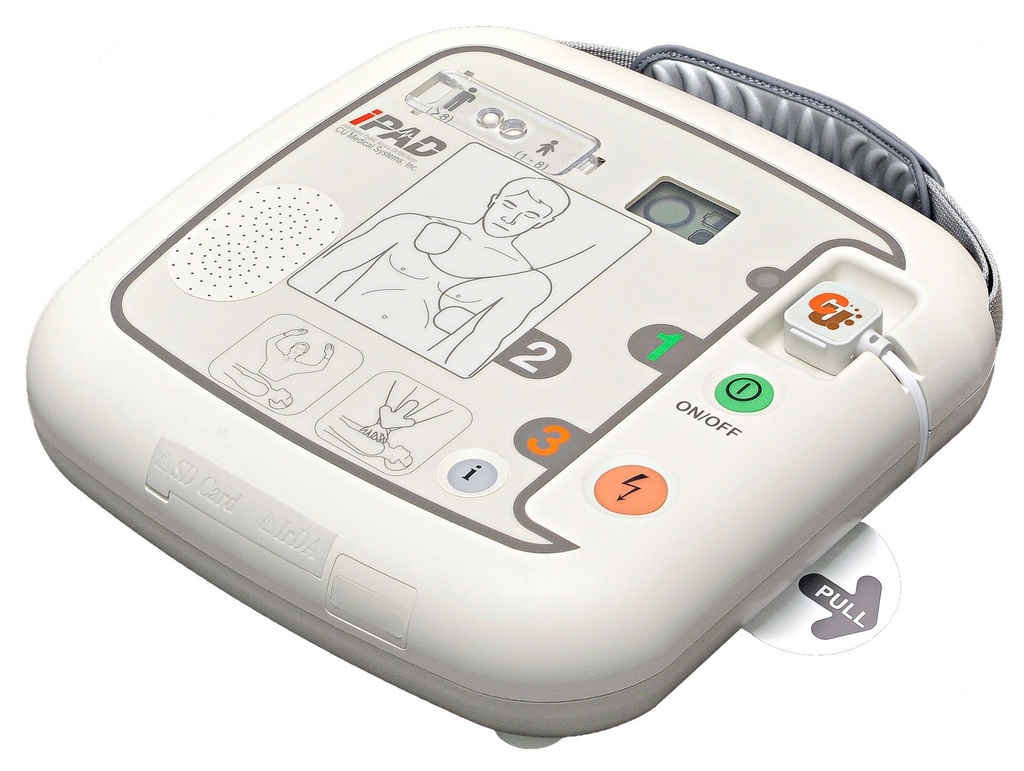
Latest-generation AED device – the semi-automatic external defibrillator iPAD CU-SP1
EASY TO UNDERSTAND
Automatic volume
Immediately after being switched on, the device detects ambient noise and automatically adjusts the volume of its voice prompts accordingly. Especially in crowded railway stations, at airports or in factory halls with lots of loud background noise, this feature can be life-saving.
READY FOR IMMEDIATE USE
Pre-connected electrodes – smartly stored
Already connected and well protected, the defibrillation electrodes are located on the underside of the device in an integrated storage compartment. This makes your AED even safer and quicker to deploy! Are the electrodes new? If used or expired “smart” electrodes are connected, the intelligent defibrillator immediately tells you what to do.
SIMPLY SORTED
Adult and paediatric mode
Is the patient a child? Thanks to a simple slider switch, you can quickly change from adult to paediatric mode. The device then works with less energy to protect the smaller heart muscle. Special paediatric electrodes are optionally available.
DOUBLE PROTECTION
Artefact and CPR detection
As an intelligent first-aid defibrillator, the CU-SP1 detects whether the patient is being touched by other people. In this case, it will not deliver a shock. Not only is the emergency patient optimally cared for – you as a first aider are also protected! In addition, the iPAD CU-SP1 detects whether you are performing chest compressions to give the emergency patient the best chance of survival.
ALWAYS IN VIEW
The clear status display
Whether battery charge level or electrode status – the LCD display lets you keep an eye on your iPAD’s functionality at all times! Is your device ready for use? Regular self-tests check that everything is in order. Do the defibrillation electrodes need to be replaced? The display warns you in good time, starting three months before the expiry date.
AFTER USE
Memory and data transfer
The iPAD CU-SP1 stores up to five emergency uses on an SD card. Would you like to evaluate the ECGs on your computer? Simply transfer the data via SD card or via the infrared interface. With the optionally available CU-EX1 software, you can edit and archive patient data.
Product data:
Defibrillator
Type: Semi-automatic external defibrillator
Model: CU-SP1
Manufacturer: CU Medical Systems, Inc.
Waveform: Biphasic truncated exponential pulse, E-cube technology
Delivered energy: 150 joules for adults, 50 joules for children, impedance-compensated
Charging time: Under 8 seconds
Controls
LCD display (electrode and battery status, correct functionality), metronome for chest compressions, voice prompts (automatic volume adjustment to the operating environment) and backlit pictograms, pre-connected electrodes, slider for adult and paediatric mode, information button for detailed voice instructions (optionally activatable),
Shock button, On/Off switch
Resuscitation guidelines
Standard preset: 5 cycles with 30 chest compressions and 2 ventilations (according to the 2010 CPR guidelines) Individual settings
Number of chest compressions, number of ventilations, number of cycles, number of chest compressions per minute, pause duration, activation of the detailed guidance (can be customised in line with future guideline changes)
Self-tests
Automatic self-diagnostic test after switching on and continuous functional tests during use; daily, weekly and monthly self-diagnostic tests Manual self-diagnostic test after each battery insertion
Environmental conditions
Operation and storage: Temperature 0 °C to 43 °C, humidity: 5% to 95% (non-condensing)
Transport: Temperature -20 °C to 60 °C
Vibration protection: Powered on: meets MIL-STD-810G, Fig. 514.6E-1, random test, Stand-by: meets MIL-STD-810G, Fig. 514.6E-2, swept sine (helicopter)
Protection class: Protected against dust and water jets, class IP55 according to DIN EN 60529: IP55
Drop protection: Withstands drops from up to 1.2 metres onto edges, corners or smooth surfaces
ESD: Complies with EN 61000-4-2:2001
EMC emissions: Meets EN 60601-1-2 limits, method EN 55011:2007 + A2:2007, Group 1, Class B
EMC immunity: Meets EN 60601-1-2 limits,
Method EN 61000-4-3:2006 + A1:2008 Level 3 (10 V/m, 80 MHz to 2500 MHz)
Event documentation
Internal memory: recording of up to 5 incidents, up to 3 hours of data Transfer via SD card or infrared interface Data analysis: PC software CU-EX1 (optionally available)
Power supply
Standard battery: 12 V DC; 2.8 Ah LiMnO2, at least 50 shocks or 4 hours of operating time, at least 3 years stand-by time
Size and weight
Dimensions: 260 mm x 256 mm x 69.5 mm
(width x length x height)
Weight: 2.4 kg (including battery and electrodes)
Kontaktinformationen im Rahmen der Produktinformationsverordnung (GPSR)
Gesetzliche Anbietererkennung:
CU Medical Germany GmbH
Berliner Str. 44, 10713, Berlin, Deutschland
Mailkontakt: